Research & Clinical Trials
If you are interested in learning more please call 913-825-2314 or 913-825-2315.

Osteoarthritis of the Knee
We are currently enrolling patients diagnosed with osteoarthritis of the knee with pain that has been present for at least 6 months. Patients must also have a history indicating insufficient pain relief from conservative treatments (medications, physical therapy, intra-articular treatment with corticosteroids) and have not had significant trauma or surgery to the knee in the past year. We are part of a multicenter clinical trial to see if these patients could benefit from an intra-articular injection into the knee.
Lumbar and Lumbosacral Radiculopathy
We are currently enrolling patients with a primary diagnosis of unilateral lumbar and/or lumbosacral radiculopathy. Patients must have had no significant improvement following a minimum of 8 weeks of mechanical intervention (physical therapy, chiropractic treatment, etc.) or over-the-counter analgesics (topical patches/creams/gels, NSAIDS). Additionally, patients cannot have a history of lumbar surgery and/or intradiscal interventions. Also, cannot have received an ESI, nerve block, or other similar procedure in the lumbosacral area performed in the past 8 weeks. We are part of a multicenter clinical trial to see if these patients could benefit from an injection in the lumbar and lumbosacral epidural space.

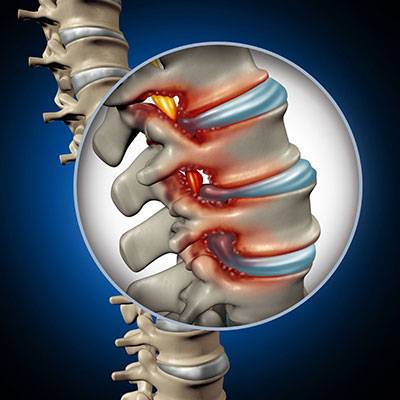
Moderate Degenerative Lumbar Stenosis
We are currently enrolling patients 45 years or older who suffer from pain, numbness, and/or cramping in their legs. Patients must have persistent leg/ buttock/ groin pain, with or without back pain, that is consistently relieved by flexion activities (such as sitting or bending over a shopping cart). We are part of a multicenter clinical trial to see if these patients could benefit from an implanted device in the lower back.
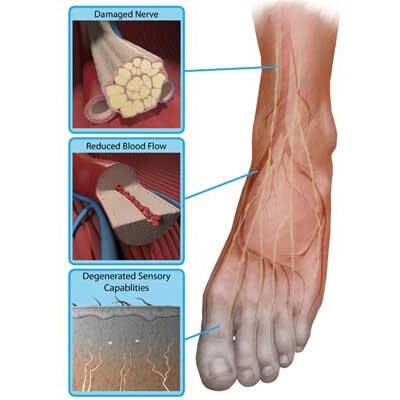
Diabetic Peripheral Neuropathy of the Feet
We are currently enrolling patients with diabetic peripheral neuropathy of the feet. This study aims to assess the long-term effectiveness of high-dose capsaicin patches for the treatment of pain associated with diabetic peripheral neuropathy.

Post-Surgical Neuropathic Pain
Neck Pain and Headache
We are currently enrolling patients with at least a 6-month history of axial neck pain and symptoms consistent with cervicogenic headache who have not had relief from conservative treatments (medications, physical therapy, chiropractic care, etc.). We are assessing whether these patients receive pain relief from Radiofrequency Ablation.


